A research team led by Qu Songnan, professor in the Institute of Applied Physics and Materials Engineering, University of Macau (UM), has proposed a strategy to prepare near-infrared (NIR) carbon nanodots by fusing large conjugated molecules. Using this strategy, the team has successfully prepared carbon nanodots with the main NIR luminescence center in aqueous solution for the first time in the world. The preparation cost of the carbon nanodots is very low, less than one percent of the cost of commercial NIR dye ICG. Moreover, the carbon nanodots are safe and non-toxic with good biocompatibility and have been successfully applied to NIR in vivo imaging and ear angiography in mouse models. This study provides new insights for the development of safe and low-cost in vivo NIR fluorescence imaging reagents. The research results have been published in the internationally renowned academic journal Advanced Science.
Compared with visible light (400-700nm), NIR light (700-1700nm) has longer wavelengths and can penetrate deeper into biological tissue, making it more suitable for biological imaging. Carbon nanodots are a new class of nano-luminescent materials, which have attracted much attention due to their low production cost, facile synthesis, and low biological toxicity. However, the emission wavelengths of the reported carbon nanodots are mainly in the visible light region, and regulating the emission bandgap of carbon nanodots has always been a research hotspot in this field. Through literature research, the research team found that the bandgap of carbon dots synthesised from small conjugated molecules such as phenylenediamine can be adjusted to the red band, but it is difficult to reach the NIR band. Theoretical calculations show that the optical bandgap of conjugated carbon dots with a particle size of about 2 nm can reach the NIR band, and most of the carbon dots reported so far exceed this size, indicating that the effective conjugation degree of these carbon dots is insufficient. Thus, it is difficult to achieve a narrow NIR emission bandgap.
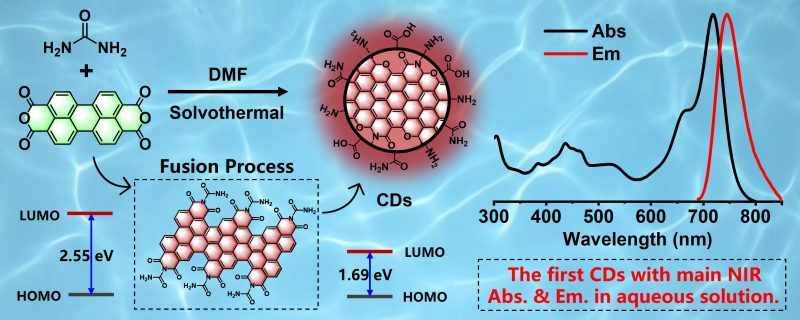
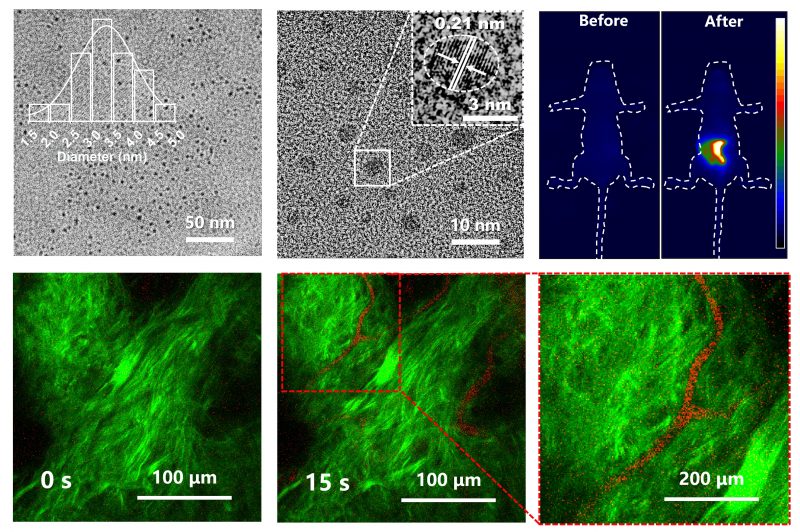
Combining theoretical calculation results, the group proposed a strategy of fusing large conjugated molecules to prepare NIR luminescent carbon nanodots. The team members condensed perylenetetracarboxylic dianhydride molecules containing five benzene rings with urea molecules through solvothermal condensation. The main absorption peak and main emission peak of the prepared carbon nanodots in the aqueous solution are both in the NIR band, and the luminescent efficiency in the NIR band is as high as 18.8 per cent, which is the highest efficiency of carbon nanodots reported so far. In the in vivo imaging experiment, the bright NIR fluorescence signal emitted from the mouse intestine after gavage for one hour showed a high signal-to-noise ratio (S/N) of 18, proving that the carbon nanodots have the ability of deep tissue NIR imaging. Furthermore, two-photon NIR FL angiography of the mouse ear was performed with two-photon fluorescence emission under 1300 nm femtosecond laser excitation. The signal of carbon nanodots (red signal) reached its highest intensity 15 seconds after injection, and blood vessels in the mouse ear were clearly visible.
Prof Qu is the corresponding author of this study. The first authors are Liu Yupeng and Wang Gang, two PhD students in the IAPME, and Lei Haipeng, a postdoctoral fellow in the Faculty of Health Sciences. This project was funded by the National Natural Science Foundation of China’s Excellent Young Scientists Fund (Hong Kong and Macao) (File no: 61922091), Science and Technology Development Fund of Macao SAR (File no: 0040/2019/A1, 0073/2019/AMJ, 0011/2019/AKP, 0128/2020/A3, 0120/2020/A3, 0026/2021/A, and 0131/2020/A3), Shenzhen-Hong Kong-Macao Science and Technology Programme (Category C) (File no: SGDX20210823103803021), and UM (CPG2020-00026-IAPME, SRG2019-00163-IAPME, MYRG2019-00103-IAPME, MYRG2020-00164-IAPME).
A schematic diagram of preparation and absorption/emission spectra of near-infrared carbon nanodots
The nanostructure of carbon nanodots, in vivo imaging in mice, and two-photon angiography of the ear
(From left) Liu Yupeng and Qu Songnan
The full version of the article can be viewed at https://doi.org/10.1002/advs.202202283
Source: Communications Office, University of Macau